
Or more of your copyrights, please notify us by providing a written notice (“Infringement Notice”) containing If you believe that content available by means of the Website (as defined in our Terms of Service) infringes one According to the Henderson-Hasselbalch equation, the pH equals the pKa if the concentration of the conjugate base equals the concentration of acid therefore, it is possible for the hydrogen ion concentration to equal the acid dissociation constant. If we have the same concentration of hydrogen ions as the acid dissociation constant ( ), then the pH will equal the pKa. This makes sense because you will have more conjugate base than acid, thereby making the solution more alkaline and increasing the pH. Increasing the ratio of to will increase the logarithm, and subsequently the pH of the solution. It is defined as:īy looking at the equation we can determine that if the ratio inside the logarithm is greater than 1, then the pH of the solution will be greater than the pKa however, if the ratio is less than 1 (meaning, if the concentration of the acid is greater than the concentration of conjugate base), then the pH will be less than the pKa. The Henderson-Hasselbalch equation is a tool that allows us to calculate the pH of an acid solution using the pKa of the acid and the relative concentrations of the acid and its conjugate base. Now we know the moles of NaOH (0.5 moles) and the concentration (2M) so we can find the volume by doing M = mol/L. This is summarized in the ICE table below. We will then be at a point where acetic acid equals acetate. If we have 1mol of acetic acid and add 0.5mol of NaOH, we will lose 0.5mol of acetic acid and gain 0.5mol of acetate. If we use the Hendersen Hasselbach equation we can see that the pH equals the pK a when the concentration of conjugate base (acetate) equals the concentration of acid. 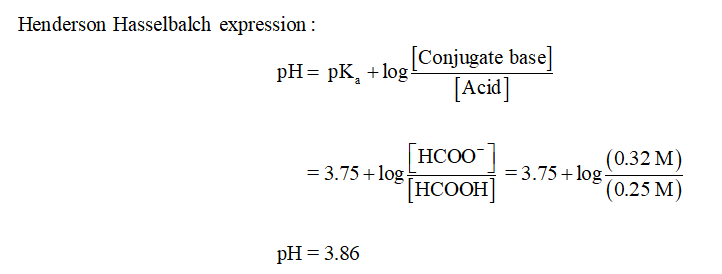
We can determine the moles of acetic acid by using M = mol/L, which gives us mol = ML = (2M) * (0.5L) = 1mol acetic acid. The reactants exist in a 1:1 ratio, so that for every mol of NaOH we add, we lose one mol of acetic acid and gain one mol of acetate. We can ignore water and sodium ions for the sake of this question. 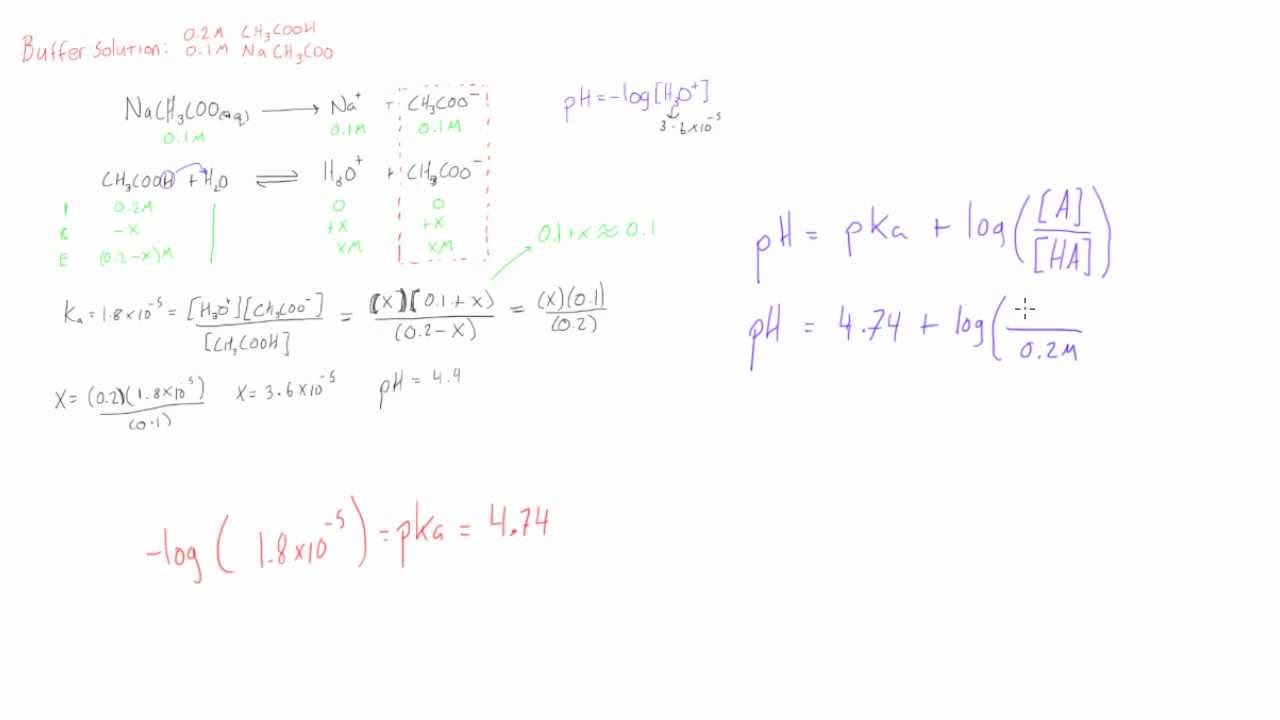
To solve this question you need to think about the chemical reaction occurring. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
